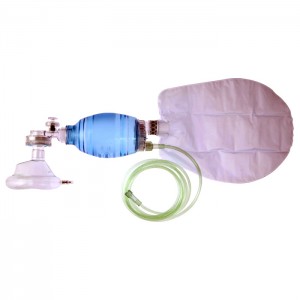
PVC Ambu Bag+ Air Cushion Mask#3 - 550ml
BV36012R / BV32045R
Maual Resuscitator Set
Designed for emergency and clinical ventilation, this resuscitator delivers stable and reliable manual ventilation.
OEM and customization options are available.
Features
- Easy to grip.
- Equipped with a pressure limitation valve for patient safety.
- A textured surface ensuring a firm grip and providing effective ventilation. Patient connector is 22/15mm.
- Conforms to the standard: ISO 10651-4:2002, and it also conforms to Council Directive MDD/93/42/EEC concerning Medical Devices.
- 100% latex free.
- It is made of phthalates free PVC.
- Squeeze resilience can be up to144 BPM per minute.
- Using high transparency material to produce, it can give doctor's more clear vision..
Specification
- Available in 3 sizes: Adult, Child and Infant
- Bag capacity: 1,600ml, 1400ml,550mml and 350ml.
- Oxygen tubing: 2 meters
- Standard patient connector: 22/15mm
- Pressure relief valve: 65cmH2O for Adult or 40cmH2O for Child and Infant models
- Catalog Show
- Related Products
PVC Ambu Bag + Pressure Release Valve+Air Cushion Mask#5 - 1600ml
BV56012R / BV52045R
Designed for emergency and clinical ventilation, this resuscitator delivers stable and reliable...
Details Add to ListPVC Ambu Bag + Air Cushion Mask#1 - 350ml
BV16012R / BV12045R
Designed for emergency and clinical ventilation, this resuscitator delivers stable and reliable...
Details Add to List- Download
Manual Resuscitator
All the material we use was made in Taiwan, we also can produce PSF type of Resuscitator, if you need this, please contact with us.
Download
Tags
How Do You Ensure Patient Safety During Pediatric Manual Ventilation in Emergency Situations?
Our PVC child resuscitator features an integrated pressure limitation valve set at 40cmH2O, specifically calibrated for pediatric patients to prevent barotrauma during emergency ventilation. Combined with a textured grip surface and high-transparency material for clear visual monitoring, this device enables your emergency team to deliver up to 144 BPM with confidence. Contact us to learn how our ISO 13485 certified resuscitator can enhance your emergency department's pediatric response capabilities.
As an ISO 13485 and FDA certified manufacturer with 28 years of experience, Omnimate Enterprise ensures comprehensive quality control following IQC, IPQC, FQC protocols and AQL 1.0 S4 standards. This 100% latex-free resuscitator set includes a 2-meter oxygen tubing and is available with OEM and customization options to meet specific clinical requirements. Our in-house EO sterilization service and well-experienced R&D team guarantee on-time delivery from prototype to series production, making us your reliable contract partner for pediatric respiratory care solutions.